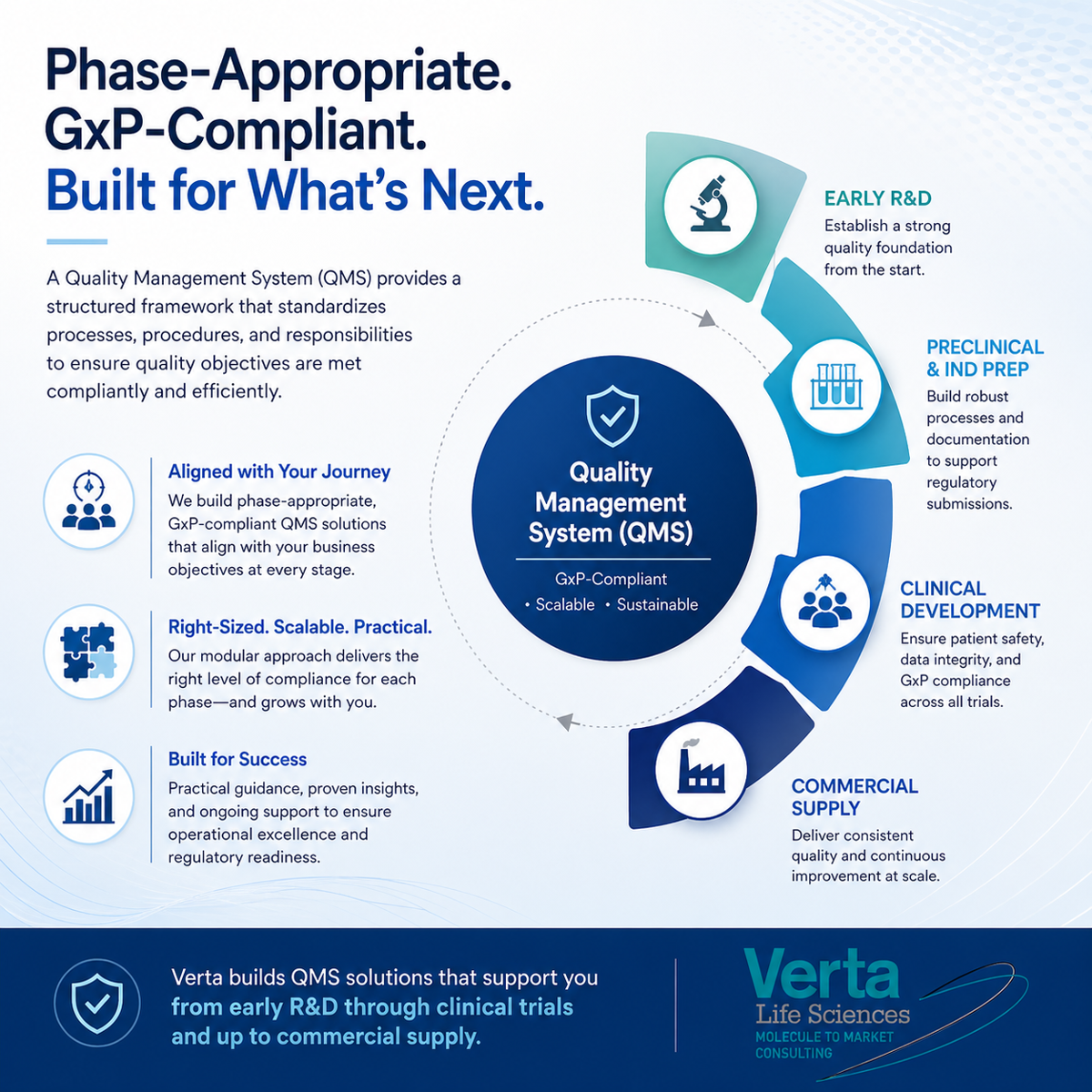
Phase-appropriate GxP-compliant QMS: 5 questions that can help
Five practical questions to help sponsors build a right-sized Quality Management System from early R&D through commercial supply.
Blog
Practical notes on CMC strategy, regulatory interactions, and manufacturing partnerships — from the team at Verta Life Sciences.

Five practical questions to help sponsors build a right-sized Quality Management System from early R&D through commercial supply.

How Verta combines in-country China QA expertise with global FDA and EMA expectations to help sponsors maintain inspection-ready supply chains.
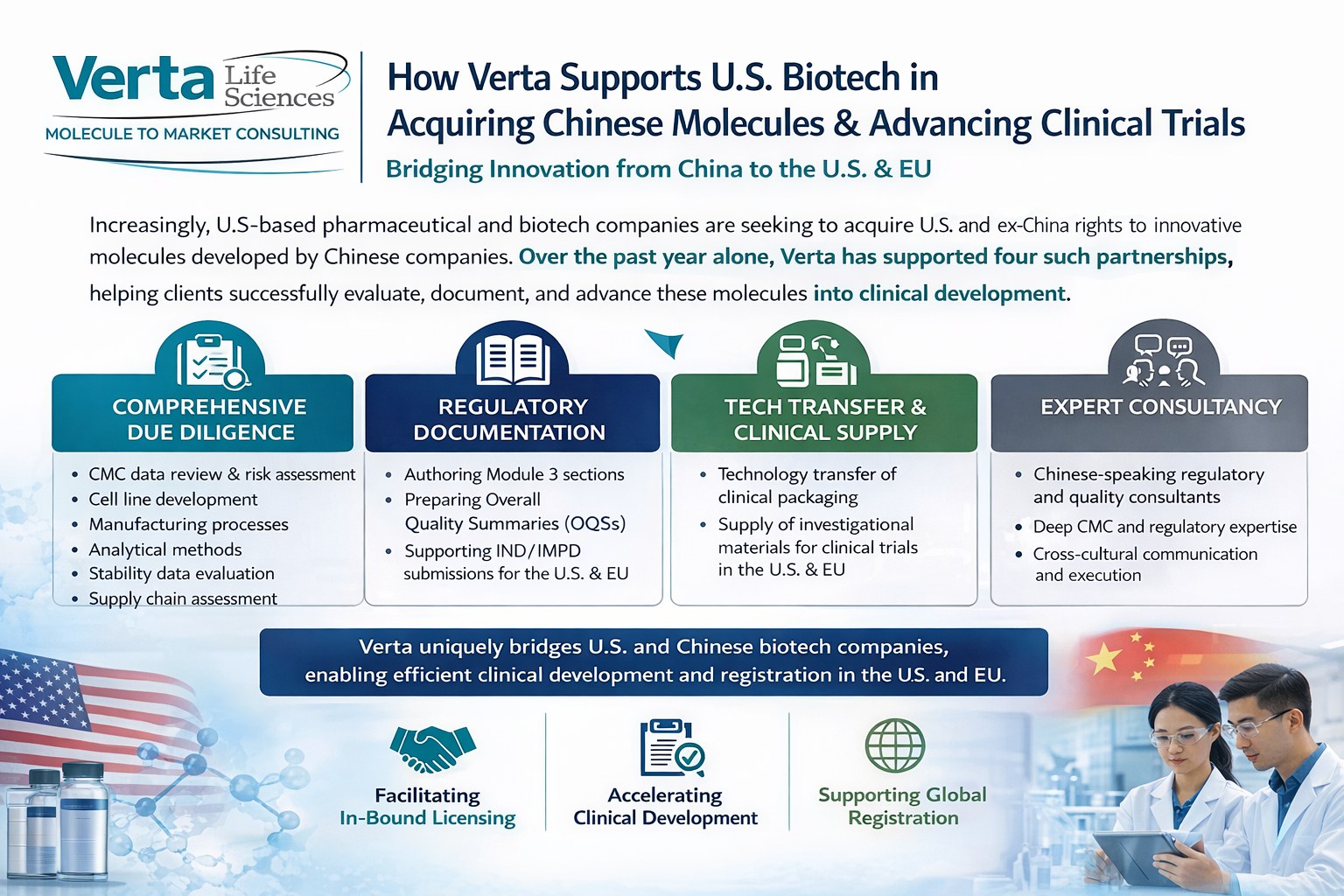
How Verta helps U.S. sponsors evaluate China-originated molecules, prepare CMC documentation, and move programs into Western clinical development.
How Verta led the entire CMC program for a monoclonal antibody COVID treatment, rapidly scaling up and managing 7 CDMOs simultaneously.
How Verta supports China due diligence for U.S. and EU sponsors - evaluating CMC components, preparing regulatory documentation, and advancing molecules into Western clinical trials.
Five practical questions to help sponsors build a right-sized Quality Management System from early R&D through commercial supply.
How Verta combines in-country China QA expertise with global FDA and EMA expectations to help sponsors maintain inspection-ready supply chains.
How Verta helps U.S. sponsors evaluate China-originated molecules, prepare CMC documentation, and move programs into Western clinical development.
A closer look at raw material sourcing, qualification, and change control — and why gaps here surface at the worst possible time.
How sponsors align validation depth with clinical stage — and where programs often fall short before commercial readiness.
A practical look at CMC-related questions, completeness, and how teams prepare for the final stretch of review.
What to verify beyond the site tour — quality systems, governance, and tech transfer readiness.