Tuesday, April 28, 2026
Phase-appropriate GxP-compliant QMS: 5 questions that can help
Verta Life Sciences
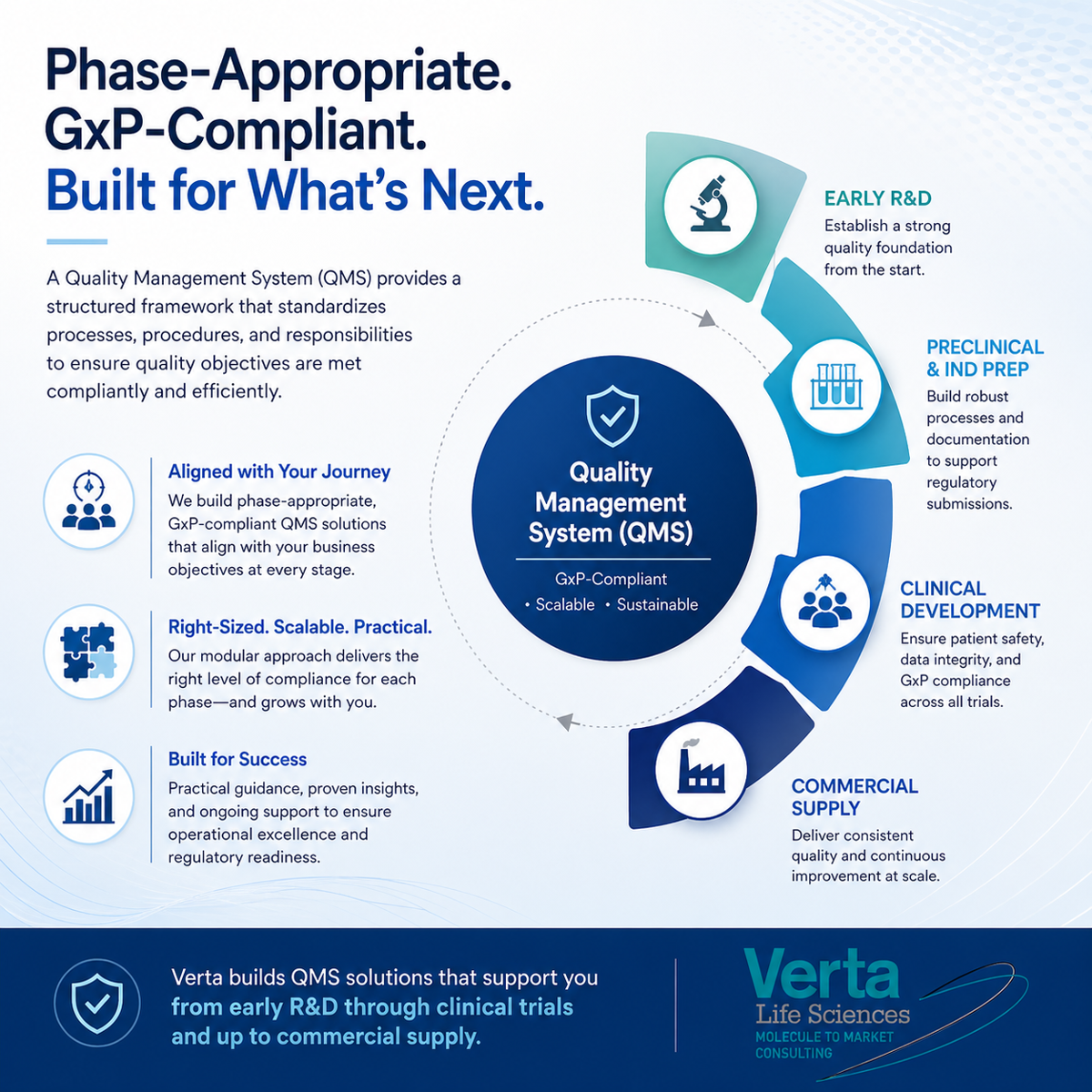
A Quality Management System (QMS) is a structured approach that standardizes procedures, processes, and responsibilities so quality objectives are met both compliantly and efficiently.
Verta helps clients develop, implement, and continually evolve a phase-appropriate, GxP-compliant QMS that aligns with business activities and objectives from early R&D through clinical development and into commercial supply. The goal is right-sized compliance at each development stage, supported by a modular QMS that can grow over time.
1. Do you understand which GxP requirements apply at each phase?
Different GxP regulations and expectations apply at different stages of drug development. Applying the right level of regulatory requirement at the right time helps balance compliance with practicality.
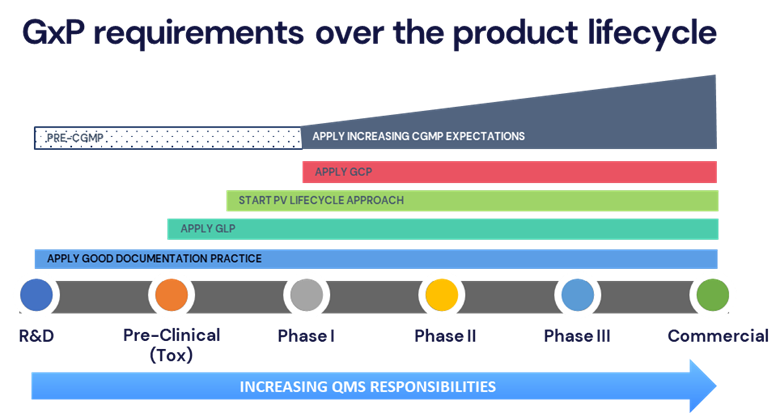
By understanding the current and future regulatory landscape, teams can implement a modular approach to QMS procedures and expand those procedures as the program moves through the development lifecycle.
2. Does your QMS scope align with your business activities?
It is easy to end up with procedures that do not clearly match the activities your organization performs. The QMS scope should include the activities you perform and enable you to operate in both a compliant and effective manner.
If certain development activities are outsourced, your own QMS does not need to duplicate every procedure for those activities. Instead, it should include robust processes for managing outsourcing, including contractor assessment, qualification, auditing, and quality agreements.
3. How can your QMS expand and adapt over time?
A scalable QMS needs a clear approach to core quality system areas, including documentation practices, data management, change management, risk management, and deviations. A consistent but evolving approach to these fundamentals helps teams move through the development lifecycle with control.
One useful step is to engage key stakeholders from each business activity area to determine how core quality elements should integrate across the organization.
4. Are your QMS procedures accurately implemented?
Procedures should use clear language and diagrams where helpful. Teams should also consider whether process steps can be built into systems that drive compliance and reduce duplication of effort.
Effective training is critical. If a common procedure applies across different business activities, such as change control, the organization needs a mechanism to maintain consistency and communicate how procedures will evolve during phased implementation.
5. Do you know if your QMS is working effectively?
As key procedures are implemented, define compliance and performance metrics that help identify trends and opportunities for improvement. Establish a consistent way to identify and communicate risks and issues so the QMS can continue to adapt.
Auditing should also be defined early. The right audit approach provides assurance of compliance while helping teams identify gaps and practical opportunities for further improvement.
Summary
A phase-appropriate GxP-compliant QMS should align with current business activities, support the next development phase, and remain practical for the people using it every day.
For more information on how Verta Life Sciences can help your business develop, implement, and evolve a compliant but efficient QMS, contact our team.